Exporting Beauty to the EU? The «Hidden» Barriers You Need to Know.
The European beauty market is one of the most lucrative in the world. But for brands outside the EU, the «Golden Gate» has many locks. At AdD Global, we’ve seen that many companies treat international expansion as a simple logistics exercise. In reality, a successful launch in Spain, the primary gateway to Europe, requires a «Legal Landing Pad.
«If you are planning to bring your skincare or cosmetics line into the EU, here is the strategic roadmap to ensure your brand arrives with precision, not friction.
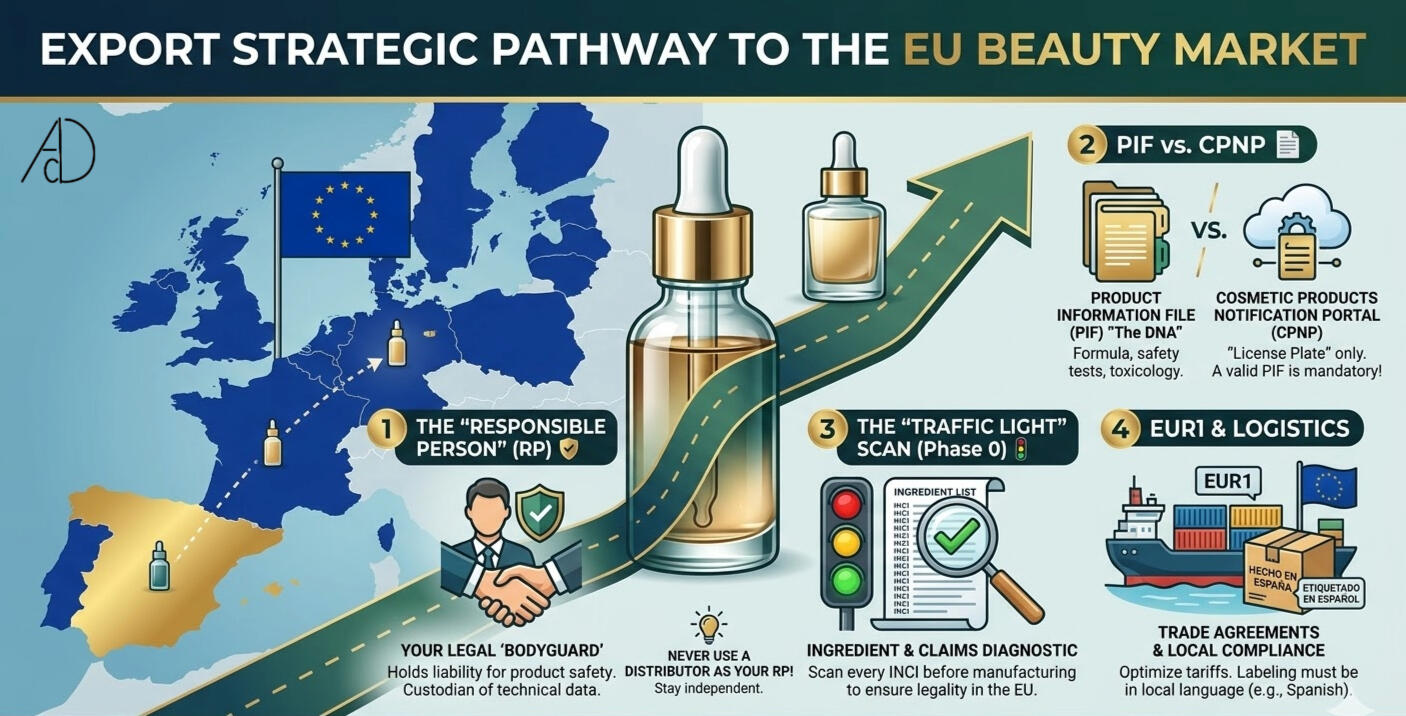
1. The «Responsible Person» (RP): Your Legal Shield 🛡️
Under Regulation (EC) No 1223/2009, you cannot sell a single unit without a legal «bodyguard» based in the EU. This is the Responsible Person (RP).
The Reality: The RP is legally liable for your product’s safety. They are the official custodian of your technical data and the sole point of contact for health authorities like the AEMPS in Spain.
Expert strategic tip: Do not appoint your distributor as your RP. If you decide to change distributors later, they effectively «own» your product registration, creating a massive operational bottleneck. Stay independent; keep your legal representation separate from your sales channels.
2. The PIF vs. The CPNP: Data over Documentation 📄
Many brands confuse «being registered» with «being compliant.
«The PIF (Product Information File): This is the technical «DNA» of your product. It includes the formula, stability tests, and toxicological assessments. It is a living document that must be ready for an immediate inspection.
The CPNP (Cosmetic Products Notification Portal): This is the notification system. Having a CPNP number is like having a license plate, thiscdoesn’t prove the car is safe to drive. Without a scientifically robust PIF, your CPNP number is simply a legal liability waiting to be discovered.
3. The «Traffic Light» Diagnostic: 🚦
The most expensive mistake in the beauty industry is printing 10,000 retail boxes before verifying that your ingredients are legal in the EU. Many «natural» or «clean» ingredients allowed in other global markets are restricted or outright banned here.
At AdD, we use a «Phase 0» running a Feasibility Scan to audit:INCI Compliance: Checking every ingredient against EU Annexes.
Product claims: Ensuring your marketing doesn’t accidentally reclassify your cosmetic as a «medicine» (a common legal red line in Spain).
4. Logistics & The EUR1 Advantage 🚢
Internationalisation isn’t just about moving boxes; it’s about cost optimization.
0% Tariffs: If your country of origin has a trade agreement with the EU, an EUR1 Certificate could allow you to import at 0% tariffs.
The labeling trap: Your labels must be in Spanish for the local market. One error in font size, ingredient naming, or the placement of the RP address, and your inventory will be blocked at the port indefinitely.
🚀 Why AdD Global?
We don’t just provide advice; we execute it for you. Our approach turns organizational complexity into your clearest competitive advantage through a structured journey:
✅ Phase 0: Ingredient Diagnostic & Claim Viability Scan.
✅ Phase 1: Regulatory & Logistics Setup. This is your roadmap to the shelf.
✅ Phase 2: Operations to automate and scale your European presence.
Bringing a beauty brand to Europe shouldn’t be a gamble. It should be a strategic, clear, and focused process.